Hedera Profiling RNA test panel
43 genes in total which includes all major ESCAT genes and all OncoKB Levels 1, 2, 3 for solid tumors.
Kits are commercialized together with the Hedera Prime software and can be purchased separately.

Fast sample-to-report workflow combining your existing instrument with Hedera Dx offering
Hands on time
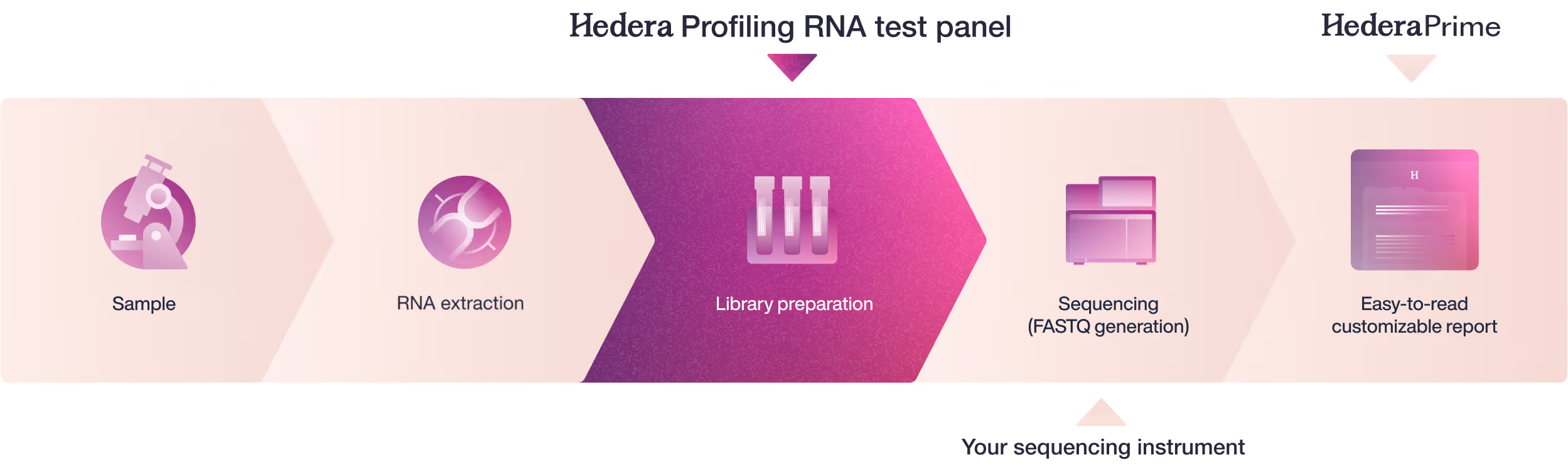
From sample to report
Tertiary analysis & reporting
Hedera Profiling RNA test panel:
Pure actionability
Compact but comprehensive
43 genes to detect fusions (partner-agnostic discovery), gene expression profiling, alternative splicing and splice variants and actionable exon skipping detection for functional insights
Highly actionable
Includes all major ESCAT genes and all OncoKB Levels 1, 2, 3 for solid tumors
Fast and accurate
Swiftly integrates into your routine NGS workflow to get accurate results within 5 calendar days, with as little as 30 ng of RNA
Robust software analysis
Backed by Hedera Prime software, save time on each report while ensuring accurate and reliable results
Hedera Profiling RNA test panel

Panel specifications
The panel offers highly sensitive and specific variant detection with 30 ng total cfDNA input, capable of identifying variants of high clinical interest, including those classified ESCAT Level I, at 0.5% VAF.
Results can be obtained within a swift 5 calendar days enabling labs to expedite testing timelines without compromising data accuracy, achieving an overall panel sensitivity > 97% and specificity > 99% at 0.5% VAF for key variants classified ESCAT Level I, across multiple cancer types.
Discover Hedera Comply, a tailored expert IVDR implementation program provided by Hedera Dx to help labs validate Hedera Profiling 2 ctDNA test panel and reach local compliance for routine testing with in-house validation and pre-filled IVDR documentation support.

Hedera Dx An implementation partner for the Lab
Comprehensive implementation program to facilitate the integration of compliant liquid biopsy in your lab.
The program provides onsite laboratory hands-on training, including a wet-lab instruction, a run assessment, a software training as well as a validation and technical support.
Supportive technical documentation is also supplied as pre-filled folder to facilitate your local compliance.

The Hedera Comply implementation program provides you with all that you need to run a local validation and develop a compliant in-house IVD liquid biopsy solution.
From onsite laboratory hands-on training to technical documentation supply and routine follow-up, Hedera Dx remains your local implementation and validation partner all the way through.
* For Research Use Only. Not for use in diagnostic procedures. This product is intended for laboratory research purposes only and is not intended for use in the diagnosis, prevention, monitoring, treatment, or alleviation of disease. This product has not been cleared or approved by any regulatory authority for clinical or diagnostic use.
1. Pascual, J et al. “ESMO recommendations on the use of circulating tumour DNA assays for patients with cancer: a report from the ESMO Precision Medicine Working Group.” Annals of oncology : official journal of the European Society for Medical Oncology vol. 33,8 (2022): 750-768. doi:10.1016/j.annonc.2022.05.520
2. Lescuyer, G et al. "Analytical Validation of a Pan-Cancer NGS Assay for In-House Liquid Biopsy Testing: An International Multicenter Study." (2024)
Manuscript in preparation. Preprint available at: https://www.medrxiv.org/content/10.1101/2024.10.17.24313324v2

