Building the Missing Real-World Clinical Evidence for Global Oncology Care
Longitudinal molecular profiling linked to clinical outcomes maps complete patient journeys, revealing how therapies perform across biomarker-defined populations, treatment lines, and different healthcare settings.

Prospective, Trial-Grade Evidence
Built on ethics committee-approved infrastructure with standardized informed consent and governance, our platform captures deep, longitudinal datasets combining clinical data, molecular profiling from tissue and liquid biopsy over time, and validated patient-reported outcomes (e.g. EQ-5D-5L, PRO-CTCAE).
These harmonized, high-quality datasets support a broad range of applications from regulatory submissions to model devlopment, covering needs for HTA and HEOR analyses, biomarker discovery, precision oncology development and clinical trial optimization including external control arms.
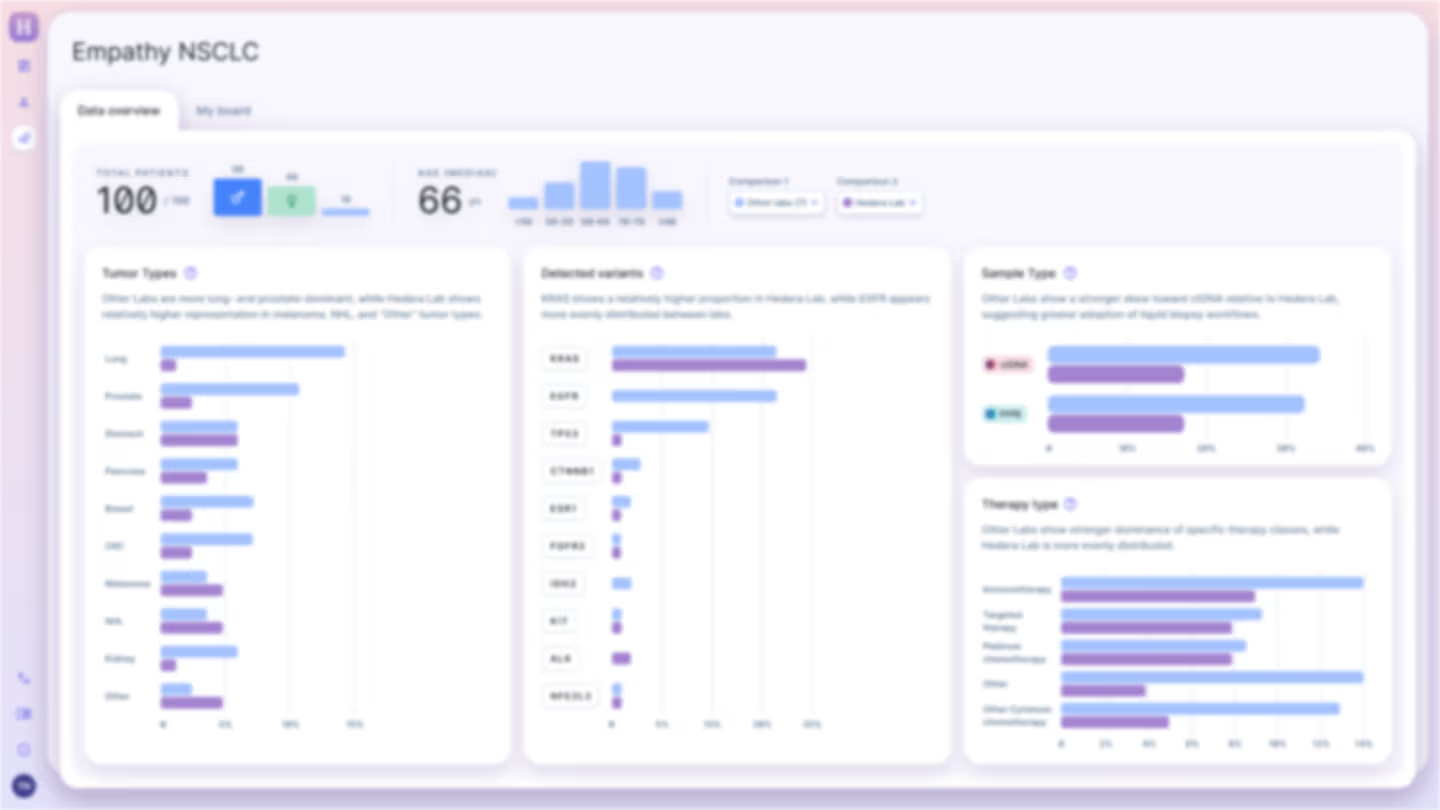
Bespoke Prospective RWE Studies
Beyond EMPATHY, we design and operate prospective studies that address specific evidence gaps in oncology. Our infrastructure ensures transparency, traceability, and regulatory-grade data quality from site activation through to analysis-ready datasets. Partners can shape study design, patient selection, biomarker panels, and endpoints, while we maintain ethical oversight and scientific independence.
