Hedera Profiling 2 ctDNA test panel
Targeted NGS ctDNA-based liquid biopsy assay for profiling of most common solid tumors including lung, breast and colorectal cancers
Kits are commercialized together with the Hedera Prime software and can be purchased separately.
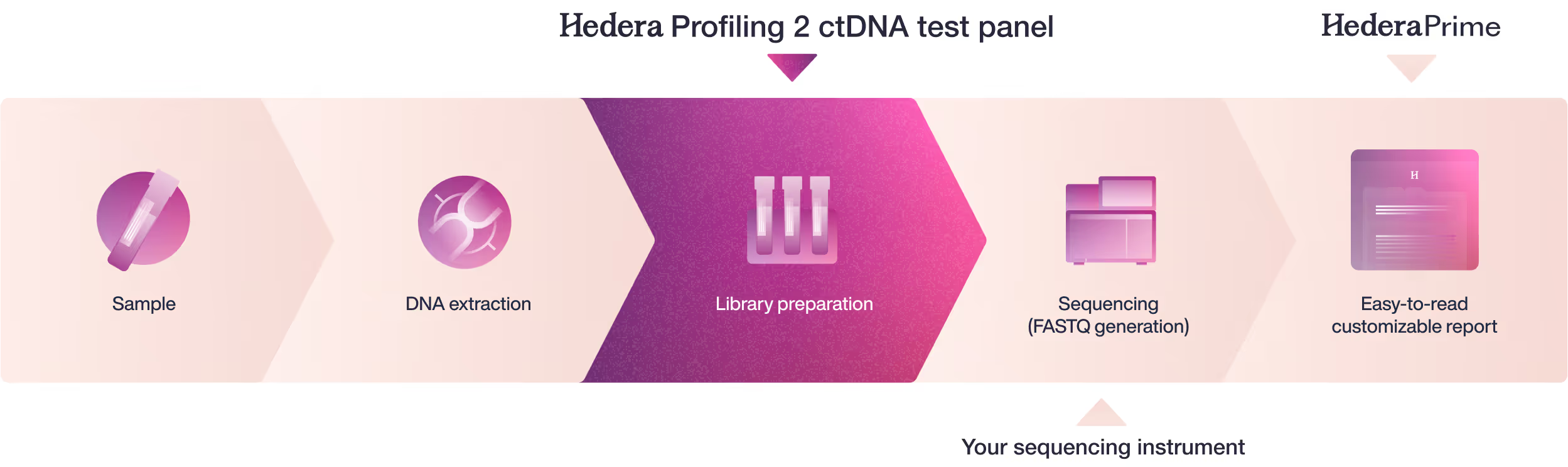
Fast sample-to-report workflow combining your existing instrument with Hedera Dx offering
Hands on time
From sample to report
Tertiary analysis & reporting
Hedera Profiling 2 ctDNA test panel:
Pure actionability
Compact but comprehensive
32 gene-panel for the detection of SNVs, Indels, CNVs, Fusions and MSI across > 10 most common tumor types in a single, DNA-only liquid biopsy assay
Highly actionable
>80% of the ESCAT Level I genes are included in the panel including ESR1, METex14 and MSI status1,2
Fast and accurate
Swiftly integrates into your routine NGS workflow to get accurate results with low input material within 5 calendar days, with as little as 10ng of cell-free DNA (30ng input recommended)
Robust software analysis
Backed by Hedera Prime software, save time on each report while ensuring accurate and reliable results
Unique fusions detection method on DNA
Cutting-edge proprietary technology to detect fusions in ctDNA with unmatched precision, enhancing the accuracy of complex variants detection in plasma samples
Hedera Profiling 2 ctDNA test panel: highly actionable for most common solid tumors
32-gene NGS panel optimized to run samples across all solid tumor types recommended by ESMO for ctDNA testing including the vast majority of ESCAT Level I biomarkers published to date
Hedera Profiling 2 ctDNA test panel includes SNVs, Indels, CNVs, Fusions, and MSI
The panel detects a broad range of cfDNA variants as well as Microsatellite Instability (MSI).

Results from a unique international (8 countries), multicenter (11 centers), analytical validation study
The panel offers highly sensitive and specific variant detection with 30 ng total cfDNA input, capable of identifying variants of high clinical interest, including those classified ESCAT Level I, at 0.5% VAF.
Results can be obtained within a swift 5 calendar days enabling labs to expedite testing timelines without compromising data accuracy, achieving an overall panel sensitivity > 97% and specificity > 99% at 0.5% VAF for key variants classified ESCAT Level I, across multiple cancer types.
Discover Hedera Comply, a tailored expert IVDR implementation program provided by Hedera Dx to help labs validate Hedera Profiling 2 ctDNA test panel and reach local compliance for routine testing with in-house validation and pre-filled IVDR documentation support.

Results based on 30 ng of input cfDNA (commercial reference control DNA) for all SNVs, Indels and Fusions with samples run on a NextSeq 2000 instrument.

137 clinical cfDNA samples were run across 9 tumor types (lung, breast, colon, prostate, pancreatic, endometrial & cervical cancers, neuroblastoma, melanoma). Results based on clinical samples run on NextSeq 500/550/550Dx and Novaseq 6000 instruments. MSI method preliminarily trained on an additional set of 54 clinical samples.
- Panel size: 90 kb
- Instruments supported: Illumina NextSeq and NovaSeq Series
- Multiplexing: up to 6 samples on a NextSeq 500/550 instrument (MID Output flow cell)
- Sample size: min 5 mL plasma required (cfDNA BCT tubes by Streck recommended for blood collection)
- cfDNA input: min 10ng cfDNA (30ng of cfDNA recommended)
- Library preparation method: hybrid-capture chemistry
- UMI/UDI technologies: allowing error correction and preventing index hopping for enhanced sensitivity and specificity
- Reactions per kit: 18
Hedera Dx An implementation partner for the Lab
Comprehensive implementation program to facilitate the integration of compliant liquid biopsy in your lab.
The program provides onsite laboratory hands-on training, including a wet-lab instruction, a run assessment, a software training as well as a validation and technical support.
Supportive technical documentation is also supplied as pre-filled folder to facilitate your local compliance.

The Hedera Comply implementation program provides you with all that you need to run a local validation and develop a compliant in-house IVD liquid biopsy solution.
From onsite laboratory hands-on training to technical documentation supply and routine follow-up, Hedera Dx remains your local implementation and validation partner all the way through.
* For performance studies only, not authorized for diagnostic procedures until its clinical performance has been established and all local regulatory requirements for routine testing are met. Not available in all countries including the United States.
$ NTRK 2/3 fusions and BRCA1/2 genes were not included in the panel to abate the overall panel size accommodating more sample multiplexing options with increased sensitivity. All other ESCAT Level I gene alterations are included.
1. Pascual, J et al. “ESMO recommendations on the use of circulating tumour DNA assays for patients with cancer: a report from the ESMO Precision Medicine Working Group.” Annals of oncology : official journal of the European Society for Medical Oncology vol. 33,8 (2022): 750-768. doi:10.1016/j.annonc.2022.05.520
2. Lescuyer, G et al. "Analytical Validation of a Pan-Cancer NGS Assay for In-House Liquid Biopsy Testing: An International Multicenter Study." (2024)
Manuscript in preparation. Preprint available at: https://www.medrxiv.org/content/10.1101/2024.10.17.24313324v2


